The welding of aluminium and its alloys
Solid solution strengthening
Very few metals are used in the pure state, as generally the strength is insufficient for engineering purposes. To increase strength the metal is alloyed, that is mixed with other elements, the type and amount of the alloying element being carefully selected and controlled to give the desired properties. An alloy is a metallic solid formed by dissolving, in the liquid state, one or more solute metals, the alloying elements, in the bulk metal, the solvent. On cooling the alloy solidifies as a solid solution which can exist over a range of compositions, all of which will be homogeneous. Depending upon the metals involved a limit of solid solubility may be
|
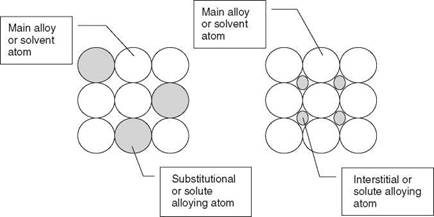
2.4 Schematic illustration of substitutional and interstitial alloying. |
reached. Microscopically a solid solution is featureless but once the limit of solid solubility is reached a second component or phase becomes visible. This phase may be a secondary solid solution, an inter-metallic compound or the pure alloying element. The introduction of a second phase results in an increase in strength and hardness, for instance iron carbide (Fe3C) in steels, copper aluminide (CuAl2) in the aluminium-copper alloys and silicon (Si) in the aluminium-silicon alloys.
In solid solution alloying the alloying element or solute is completely dissolved in the bulk metal, the solvent. There are two forms of solid solution alloying - interstitial and substitutional - illustrated in Fig. 2.4. Interstitial alloying elements fit into the spaces, the interstices, between the solvent atoms, and substitutional elements replace or substitute for the solvent atoms, provided that the diameter of the substitutional atom is within ±15% of the solvent atomic diameter. The effect of these alloying elements is to distort the space lattice and in so doing to introduce a strain into the lattice. This strain increases the tensile strength but as a general rule decreases the ductility of the alloy by impeding the slip between adjacent planes of atoms.
Many elements will alloy with aluminium but only a relatively small number of these give an improvement in strength or weldability. The most important elements are silicon, which increases strength and fluidity; copper, which can give very high strength; magnesium which improves both strength and corrosion resistance; manganese, which gives both strength and ductility improvements; and zinc, which, in combination with magnesium and/or copper, will give improvements in strength and will assist in regaining some of the strength lost when welding.
Property
|
Amount of cold work |
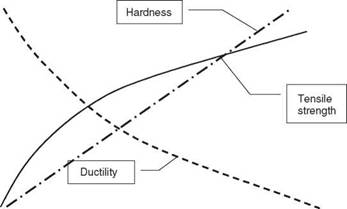
2.5 Illustration of the effect of cold work on strength, hardness and ductility.