The welding of aluminium and its alloys
Aluminium weldability problems
2.3.1 Porosity in aluminium and its alloys
Porosity is a problem confined to the weld metal. It arises from gas dissolved in the molten weld metal becoming trapped as it solidifies, thus forming bubbles in the solidified weld (Fig. 2.7).
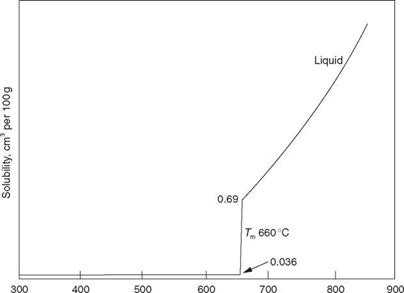
Porosity can range from being extremely fine micro-porosity, to coarse pores 3 or 4 mm in diameter. The culprit in the case of aluminium is hydrogen, which has high solubility in molten aluminium but very low solubility in the solid, as illustrated in Fig. 2.8. This shows a decrease of solubility to the order of 20 times as solidification takes place, a drop in solubility so
|
2.7 Finely distributed porosity in TIG plate butt weld 6mm thickness. Courtesy of TWI Ltd. |
|
Temperature, °C 2.8 Solubility of hydrogen in aluminium. |
pronounced that it is extremely difficult to produce a porosity-free weld in aluminium.
Porosity tends to be lowest in autogenous welds. When filler metal is used porosity levels tend to increase because of contamination from the wire. Of the conventional fusion welding processes TIG has lower levels of porosity than MIG due to this hydrogen contamination of the wire. Increasing the arc current increases the temperature of the weld pool and thereby increases the rate of absorption of hydrogen in the molten metal. Conversely, in the flat welding position increasing the heat input can reduce porosity when the rate of gas evolution from the weld exceeds the rate of absorption - slowing the rate at which the weld freezes allows the
hydrogen to bubble out of the weld. A similar effect can be achieved by reducing the travel speed. Increasing arc voltage and/or arc length increases the exposure of the molten metal to contamination, and porosity will thereby increase. The alloy composition can also influence the amount of porosity by changing the solubility of hydrogen - magnesium in particular has a beneficial effect. It is thought that magnesium raises the solubility and reduces absorption of hydrogen by as much as twice at 6% Mg. Copper and silicon have the opposite effect. A conclusion that can be drawn from this is that when porosity is encountered the use of Al-Mg filler can assist in reducing the problem. This assumes of course that such filler metal is acceptable in the specific application.
The sources of hydrogen are many and varied but one of the primary sources is the welding consumables. Moisture is an intrinsic part of the flux in any of the flux shielded processes such as manual metallic arc (MMA) or SMA (shielded metal arc), and submerged arc (SA) welding. During welding this moisture decomposes in the arc to give hydrogen, resulting in a large amount of porosity. This is one reason why these processes are not widely used to weld aluminium.
The gas used in the gas shielded processes is another source of moisture which is easy to overlook. Ideally gas with a dew point of less than -50 °C (39 ppm water) should be used. To achieve such a high purity it is essential to purchase the gas with a guaranteed low dew point. It is also necessary to ensure that when it is delivered to the weld pool it has maintained this high degree of purity. This means that the gas supply system should be checked at regular intervals for leaks, that damaged hoses are replaced immediately and joints are sound. When faced with a porosity problem the gas purity should be checked first of all at the torch nozzle before working back along the gas delivery system in a logical manner to locate the source of contamination. If the workshop layout permits it is recommended that the gas is supplied from a bulk tank rather than from cylinders and distributed around the workplace in copper or steel piping. Despite the best efforts of the gas suppliers it is not always possible to guarantee completely the purity of individual bottles except at great expense. Bulk supplies are generally of superior quality. Screwed or bolted flanged connections are potential sources of contamination and leaks and are best avoided by the use of a brazed or welded system.
A further source of contamination may come from the gas hoses themselves. Many of the plastics used for gas hoses are porous to the water present in the air. This results in moisture condensing on the inside of the hose and being entrained in the shield gas. A number of reports published recently have identified the permeability of hose compositions and a summary of the results is presented in Table 2.2. From this it can be seen that only a limited number of hose compositions will maintain gas purity.
|
Table 2.2 Moisture permeability of gas hoses
|
Of the plastic tubing the most porous is neoprene rubber, the least porous polytrifluoro-chloroethylene. The best of all is an all-metal system. Any plastic hoses should be kept as short and as small a diameter as possible consistent with the application.
Also important is the fact that the moisture collects in the tube over a period of time when no gas is flowing. The implication of this is that if welding equipment is left idle for long periods of time the first few welds to be made on recommencing welding may contain unacceptable porosity. A systematic porosity problem always occurring, for example, at the commencement of the first shift after a weekend break may be an indication of this problem. Flushing the hoses through for a short time by operating the torch trigger may help to reduce the amount of porosity. If this is done with the MIG (GMAW, gas metal-arc welding) torch do not forget to slacken off the wire drive rolls!
TIG welding wire should be cleaned with a lint-free cloth and a good degreasant before use. Once the wire has been cleaned do not handle the wire with bare hands but use a clean pair of gloves, store the wire in clean conditions and weld within a short time of cleaning. For the MIG process there are devices available that can be fitted around the wire where it enters the torch liner in the wire feed unit and that will clean the wire as it passes through. Best of all the wire should be shaved to remove any contaminants and oxides that may have been pressed into the surface during the wire drawing operation.
Cleanliness of the parent metal is also extremely important in achieving low levels of porosity - it cannot be emphasised too strongly how important this is. Thorough degreasing is essential, followed by a mechanical cleaning such as stainless steel wire brushing to remove the oxide layer which may be hydrated. Once degreased and wire brushed the parent material should be welded within a short period of time, a period of four hours frequently being regarded as acceptable. Further details of mechanical cleaning, degreasing and workshop conditions are given in Chapter 4.
22 The welding of aluminium and its alloys Table 2.3 Summary of causes and prevention of porosity
|
Remedial measures |
|
Potential causes |
Mechanism of porosity formation
Hydrogen
entrapment
Gas/air
entrapment
Rapid freezing trapping gas
Erratic wire feed
Oxide film, grease, drawing soap on filler wire; oxides, grease, dirt on parent plate; dirt/grease in liner; contaminated shield gas; water leaks in torch; spatter on weld face.
(a) Weld pool turbulence due to high current.
(b) Gas expanding from root of partial penetration/fillet welds.
Heat input too low, rapid heat loss, viscous weld pool, cold backing bar.
Kinked, blocked or wrong size liner, incorrect or badly adjusted drive rolls, damaged contact tip, unstable power supply.
Clean wire, use high - quality gas, change liner, protect wire from contamination, change torch, clean plate, minimise spatter.
(a) Use lower current, reduce travel speed, change gun angle.
(b) Use full pen weld, allow gap in fillet weld root, use high heat input.
Increase current, slow travel speed, consider preheat, heat backing bar, replace argon shield gas with helium.
Straighten wire conduit, replace contact tip, adjust drive roll pressure, fit correct liner, fit grooved rolls.
A last source of porosity may be hydrogen dissolved within the aluminium. Although solubility of hydrogen is low in the solid phase there can be sufficient in the parent metal to give a problem on welding. This is unlikely in wrought products but may arise when welding castings or sintered products. For this reason some purchasers specify in their purchase orders a limit on hydrogen, typically 2 ppm. Avoidance of porosity when hydrogen is present in the parent metal is impossible to avoid.
Table 2.3 summarises the causes and prevention of porosity.