Solar energy in progress and future research trends
Sun radiation
Earth receives virtually all of its energy from space in the form of solar electromagnetic radiation. Its total heat content does not change significantly with time, indicating
a close overall balance between absorbed solar radiation and the diffuse stream of low-temperature thermal radiation emitted by the planet. The radiance at the mean solar distance—the solar constant—is about 1360 W/m2 [86]. The solar radiation varies according to the orbital variations. If Ss is the total solar radiation output from the sun in all frequencies then at a distance R from the sun-center, the flux of the radiation will be the same assuming that the radiation is equal in all directions. If the radiation flux per unit area at a distance R is represented by Q(R), then the total radiation is equal to 4pR2Q(R). Hence, it is possible to write that
Ss = 4pR2 Q(R) (1)
or
Q(R) = Ss /4pR2 (2)
The earth is approximately 150 X 106 km away from the sun. Hence, Eq. (2) yields approximately that the total solar output is about 3.8 X 1026 W. Of course, the radiation
incident on a spherical planet is not equal to the solar constant of that planet. The earth intercepts a disk of radiation from the sun with area pr2, where r is the radius of the earth. Since, the surface area of the earth is 4pr2, the amount of solar radiation per unit area on a spherical planet becomes as
pR2Q(R)/4pr2 = Q(R)/4 (3)
Consequently, the average radiation on the earth’s surface can be calculated as 1360/4 = 340 W/m2. All these calculations assume that the earth is perfectly spherical without any atmosphere and revolves on a circular orbit. Of course, these simplifications must be released in practical applications.
The driving force for the atmosphere is the absorption of solar energy at the earth’s surface. Over time scales long compared to those controlling the redistribution of energy, the earth-atmosphere system is in the thermal equilibrium. The absorption of solar radiation, at visible wavelengths as short-wave (SW) radiation, must be balanced by the emission to space of infrared or long-wave (LW) radiation by the planet’s surface and atmosphere. A simple balance of SW and LW radiations leads to equivalent blackbody temperature for earth at T = 255 K. This is some 30 K colder than the global-mean surface temperature Ts < 288 K. The difference between these two temperatures follows from the greenhouse effect, which results from the different ways the atmosphere processes SW and LW radiations. Although transparent to SW radiation (wavelength < 1 mm), the same atmosphere is almost opaque to LW radiation (wavelength < 10 mm) reemitted by the planet’s surface. By trapping radiant energy that must eventually be rejected to space, the atmosphere’s capacity elevates the surface temperature over what it would be in the absence of an atmosphere. From the time it is absorbed at the surface until it is eventually emitted to space, energy assumes a variety of forms.
The oscillating field plane of electric and magnetic waves are perpendicular to each other, i. e. when the electric field E, and magnetic field Hm are in the yz-plane, respectively, the propagation direction is along the x-axis. Solar radiation electromagnetic waves travel with the speed of light and cover the distance between the sun and the earth at about 8 min. The wavelength l of the wave is related to the frequency n through the light velocity, c, as
c = In (4)
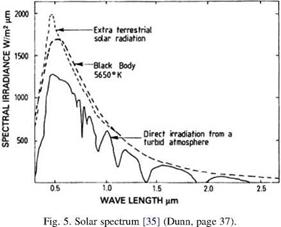
Solar energy spectrum contains wave lengths, which are too long to be seen by naked eye (the infra-red), and also wave lengths, which are too short to be visible (the ultraviolet). The spectral distribution of the solar radiation in W/m2 per micrometer of wavelength; that is, it gives the power per unit area between the wavelength range of l and l + 1, where l is measured in micrometers. The solar spectrum is roughly equivalent to a perfect black body at a temperature of 5800 K. After the combined effects of water
|
|
vapor, dust, and adsorption by various molecules in the air, certain frequencies are strongly absorbed and as a result the spectrum received by the earth’s surface is modified as shown in Fig. 5. The area under the curve gives the total power per square meter radiated by a surface at the specified temperature. The earth receives its radiation from the sun at short wavelength around a peak of 0.5 mm, whereas it radiates to space at a much lower wavelength around a peak value of 10 mm, which is well into the infrared. The relationship between the wavelength Amax, which is the power radiated is a maximum, and its relationship with the body temperature is given as Wien’s law which reads as [21,40]
1maxT = 3 X 10"3 mK (5)
Electromagnetic waves show particle properties as photons, and in particular, they behave as if they were made up of energy packets, having an energy E, which is related to frequency n as,
E = hn (6)
where h is the universal Plank constant, h = 6.626 X 10234 J s.
It is well known by now that the planets, dusts, and gases of the solar system orbit around the enormous central sun contains 99.9% of the mass of the system and provides the gravitational attraction that holds it together. The average density of the sun is slightly greater than of water as 1.4 g/cm3. One of the reasons for sun’s low density is that it is composed predominantly of hydrogen, which is the slightest element. Its massive interior is made up of matter held in gaseous state by enormously high temperatures. Consequently, in smaller quantities gases would rapidly expand and dissipate at such extreme temperatures. The emitted energy of the sun is 3.8 X 1026 W and it arises from the thermonuclear fusion of hydrogen into helium at temperatures around 1.5 X 106 K in the core of the sun which is given by the following chemical equation
4jH!3 4He + 2b + energy(26.7 MeV)
In the sun’s core, the dominant element is helium (65% by mass) and the hydrogen content is reduced to 35% by mass as a direct result of its consumption in the fusion reactions. It is estimated that the remaining hydrogen in the sun’s core is sufficient to maintain the sun at its present luminosity and size for another 4 X 109 years. There is a high-pressure gradient between the core of the sun and its perimeter, which is balanced by the gravitational attraction of the mass of the sun. The energy released by the thermonuclear reaction is transported by energetic photons, but because of the strong adsorption by the peripheral gases most of these photons do not penetrate the surface. In all regions of the electromagnetic spectrum the outer layers of the sun continuously lose energy by radiation emission into space in all directions. Consequently, a large temperature gradient exists between the core and the outer parts of the sun.
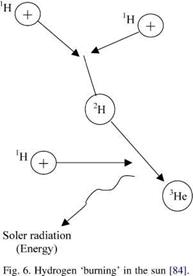
Until the rise of modern nuclear physics, there was no known source for the sun’s energy, but it is now clear that the solar interior is a nuclear furnace that releases energy in much the same way as do man-made thermonuclear - explosions. It is by now obvious through spectroscopic measurements of sunlight reaching the earth from the photosphere layer of the sun that the solar mass is composed predominantly of the two lightest elements—hydrogen, which makes up about 70%, and helium about 27%, and the remaining 3% of solar matter is made up of all the other 90-odd elements [84]. The origin of solar irradiation that is received on the earth is the conversion of hydrogen into helium through solar fusion. Theoretical considerations show that at the temperatures and pressures of the solar interior, helium is steadily being produced from lighter hydrogen as four nuclei unite to form one nucleus of helium as presented in Fig. 6. During such a conversion single hydrogen nuclei (proton) made unstable by heat and pressure first combine to form double hydrogen nuclei
|
|
which then unite with a third hydrogen nucleus to form helium-3, with a release of electromagnetic energy. The helium nucleus formed in this fusion possesses weights slightly less than the combined weight of the four hydrogen atoms, which gave rise to it. This small excess of matter is converted directly to electromagnetic radiation and is the unlimited source of solar energy.
The source of all renewable energy is the enormous fusion reactor in the sun, which converts hydrogen into helium at the rate of 4 X 106 tonnes/s. Sun’s surface temperature is approximately 6000 °C and it radiates electromagnetic energy in terms of photons, which are light particles. Almost one-third of this incident energy on the earth is reflected back, but then the rest is absorbed, and eventually retransmitted to deep space in terms of long-wave infrared radiation. Today, the earth radiates just as much energy as it receives and sits in a stable energy balance at a temperature suitable for life on the earth. In fact, solar radiation is in the form of white light and it spreads over a wider spectrum of wavelengths from the short-wave infrared to ultraviolet. The wavelength distribution is directly dependent on the temperature of the sun’s surface.
As the solar radiation reaches the upper boundary of earth’s atmosphere, the light starts to scatter depending on the cloud cover and the atmospheric composition [56]. A proportion of the scattered light comes to earth as diffuse radiation. The term ‘sunshine’ implies not the diffuse but direct solar radiation that comes straight from the sun. On a clear day, direct radiation can approach a power density of 1 kW/m2, which is known as solar power density for the solar collector testing purpose (Section 11).
All solid, liquid and gaseous matters are no more than a vibrating cosmic dance of energy, which can be perceived by human in three-dimensional form, structure, density, color and sound. Density makes the matter as solid, liquid or gaseous in addition to the movement of its atoms, molecules also give rise to the sensations of heat and cold. The interaction of matter with the area of the electromagnetic spectrum, which is known as light gives it color, which is perceived through the eyes. However, if one takes a step inwards it can be observed that matter is composed of large and small scale molecules. Each atom until the advent of modern physics was considered to consist of a nucleus of positively charged protons and zero-charged neutrons, with a number of ‘shells’ of orbiting negatively charged electrons. With these particles the hydrogen and helium atoms are shown in Fig. 7. In modern physics the subatomic particles are considered as wave packets as electromagnetic force fields and as energy relationships. They have ‘spin’ and they rotate about the axis of their movement. They have no ‘oscillation’, like an ultra-high-speed-pendulum. Whilst spinning and oscillating they move around relative to each other in three dimensions. They also have an ‘electrical charge’ and a ‘magnetic moment’ and therefore, an ‘electromagnetic field’.
|
|
An account of the earth’s energy sources and demand cannot be regarded as complete without a discussion of the sun, the solar system and the place of the earth within this system. In general, the sun supplies the energy absorbed in short terms by the earth’s atmosphere and oceans, but in the long terms by the lithosphere where the fossil fuels are embedded. Conversion of some of the sun’s energy into thermal energy derives the general atmospheric circulation [14]. A small portion of this energy in the atmosphere appears in the form of kinetic energy of the winds, which in turn derive the ocean circulation. Some of the intercepted solar energy by the plants is transformed virtually by photosynthesis into biomass. In turn, a large portion of this is ultimately converted into heat energy by chemical oxidation within the bodies of animals and by the decomposition and burning of vegetable matter. On the other hand, a very small proportion of the photosynthetic process produces organic sediments, which may eventually be transformed into fossil fuels. It is estimated that the solar radiation intercepted by the earth in 10 days is equivalent to the heat which would be released by the combustion of all known reserves of fossil fuels on earth.
The total power that is incident on the earth’s surface from the sun every year is 1.73 X 1014kW and this is equivalent to 1.5 X 1018 kW h annually, which is equivalent to 1.9 X 1014 cet. Compared to the annual world consumption of almost 1010 cet, this is a very huge and unappreciable amount. This is approximately about 10,000 times greater than what is consumed on the earth annually. By engineering considerations this energy is considered as uniformly spread all over the world’s surface, and hence the amount that falls on 1 m2 at noon is about 1 kW in the tropical regions. This solar power density varies with latitude, elevation and season of the year in addition to time in a particular day. Most of the developing countries lie within the tropical belt of the world where there are high solar power densities, and consequently, they want to exploit this source in the most beneficial ways. On the other hand, about 80% of the world’s population lives between latitudes 35°N and 35°S. These regions receive sun radiation for almost 3000-4000 h per year. In solar power density terms, this is equivalent to around 2000 kW h/yr again cet as
0. 25. Additionally, in these low latitude regions seasonal sunlight hour changes are not significant. This means that these areas receive sun radiation almost uniformly throughout the whole year.
Apart from the solar radiation the sunlight also carries energy. It is possible to split the light into three overlapping groups as:
1. photovoltaic group: produces electricity direct from the sun’s light,
2. photochemical group: produces electricity, or, light and gaseous fuels by means of non-living chemical processes, and finally,
3. photobiological group: produces food (animal and human fuel) and gaseous fuels by means of living organisms or plants.
The last two groups also share the term ‘photosynthesis’ This means literally the building (synthesizing) by light.